 Thus, the molecule with the maximum dipole moment among CO2, CH4, NH3, and NF3 is NF3. Therefore, NF3 has a higher dipole moment than NH3. Since fluorine is more electronegative than nitrogen, the N-F bonds in NF3 are more polar than the N-H bonds in NH3. However, to determine which molecule has the maximum dipole moment between NH3 and NF3, we need to consider the electronegativity difference. Thus, NF3 has a dipole moment.Īmong the given molecules, NH3 and NF3 have dipole moments, while CO2 and CH4 have zero dipole moments. The dipole moments of the N-F bonds do not cancel each other out, resulting in a net dipole moment. The molecule is also unsymmetrical due to the presence of the lone pair on nitrogen. Fluorine is highly electronegative, and nitrogen is moderately electronegative. NF3 has a trigonal pyramidal molecular geometry similar to NH3, with the nitrogen atom at the center and three fluorine atoms surrounding it. The lone pair causes a distortion in the electron cloud around the nitrogen atom, resulting in a net dipole moment. The molecule is also unsymmetrical due to the presence of the lone pair. Nitrogen is more electronegative than hydrogen, creating polar N-H bonds. NH3 has a trigonal pyramidal molecular geometry with the nitrogen atom at the center and three hydrogen atoms and a lone pair of electrons surrounding it. 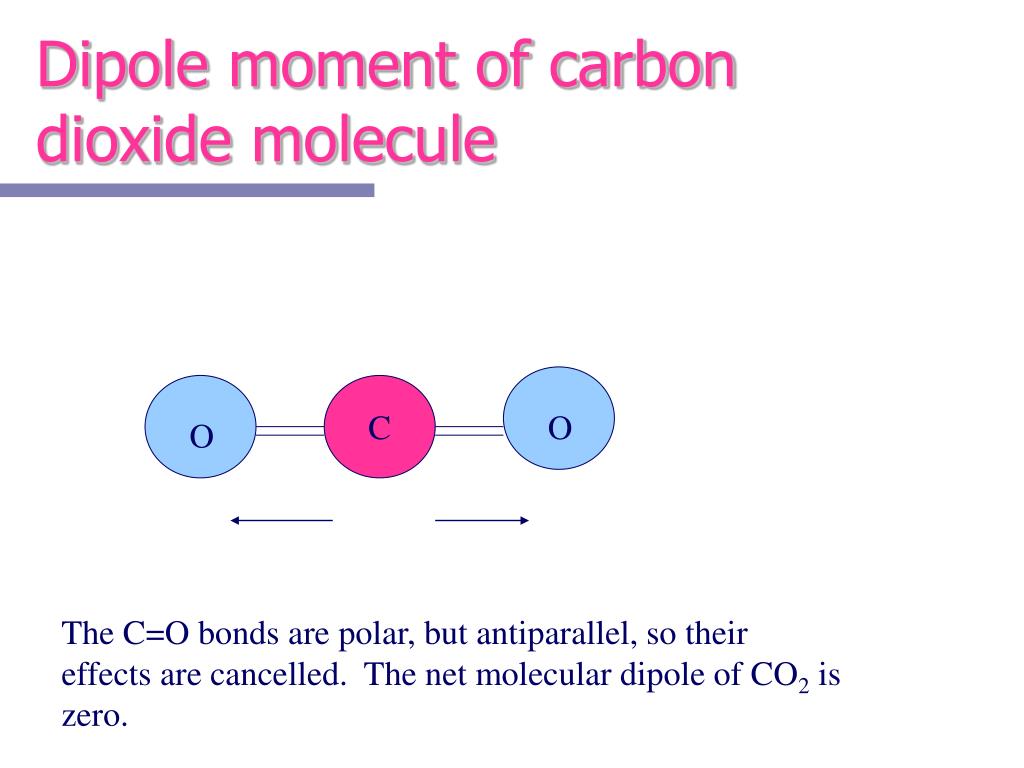
Additionally, the molecule is symmetrical, with all bond dipoles pointing towards the center, thereby canceling out each other. Carbon and hydrogen have similar electronegativities, so the C-H bonds are nonpolar. Therefore, CO2 has zero dipole moment.ĬH4 has a tetrahedral molecular geometry with the carbon atom at the center and four hydrogen atoms surrounding it. However, the molecule as a whole is nonpolar because the bond dipoles cancel each other out. Oxygen is more electronegative than carbon, resulting in a polar bond between carbon and each oxygen atom. It depends on the electronegativity difference between the atoms and the molecular geometry.ĬO2 has a linear molecular geometry due to its central carbon atom being bonded to two oxygen atoms. Dipole moment is a vector quantity that measures the separation of positive and negative charges in a molecule. Their vector sum is zero, so CO2 therefore has no net dipole. To determine the molecule with the maximum dipole moment among CO2, CH4, NH3, and NF3, we need to evaluate the polarity of each molecule. (a) In CO2, the CO bond dipoles are equal in magnitude but oriented in opposite directions (at 180°). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
